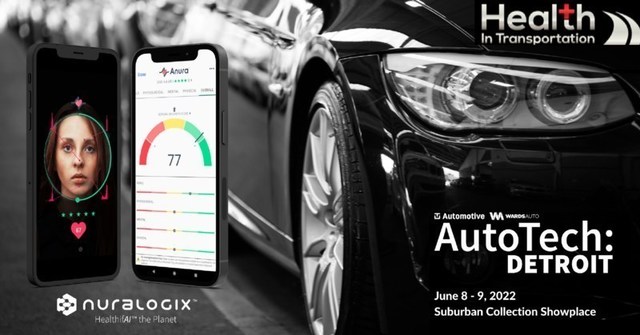
DETROIT, June 8, 2022 — Professional drivers, working both in mass transit and in trucking, are now able to access a 30-second CDL health check that focuses on the…
First AI to Refine Medical Coding by Exploring Therapeutic Data
SAN FRANCISCO, June 2, 2022 — Medical AI start-up Aesop Technology announced a new partnership that made their new product, DxPrime, available in the Olive Library. DxPrime provides physicians and clinical…
Panoramic Atlases of Life Published in Cell Mark Establishment of SpatioTemporal Omics Consortium
STOC launches with more than 80 scientists joining globally, focused on obtaining the greatest scientific benefits from spatially resolved transcriptomics technology; first studies use BGI-Research’s Stereo-seq to produce spatiotemporal…
Dada’s JDDJ in action to guarantee medical supply and online health consultation with pharmaceutical partners
SHANGHAI, April 2, 2022 — As a number of cities in China are ramping up anti-epidemic measures, more consumers rely on online-to-offline services to purchase much-needed medications. JDDJ, one of…
Breakthrough Study Validates Artificial Intelligence as a Novel Biomarker in Predicting Immunotherapy Response – Published in Journal of Clinical Oncology
Lunit’s latest study provides evidence for Lunit SCOPE IO’s ability to predict immunotherapy response This is the first study that adopted…
Happiness Development to Acquire Hekangyuan, One of the Largest Healthcare Product Distributors in Fuzhou Area in China
NANPING, China, March 7, 2022 — Happiness Development Group Limited ("HAPP" or the "Company"), (NASDAQ: HAPP) an emerging and diversified company engaging in the business of production of nutraceutical and dietary supplements,…
China.org.cn: Hong Kong’s fight against COVID-19: a test for “one country, two systems”
BEIJING, March 4, 2022 — A news report from China.org.cn on Hong Kong’s COVID-19 pandemic situation:   Hong Kong has been taken aback by the 5th onset of…
Vocera, now part of Stryker, introduces latest evolution in hands-free communication to help protect and connect healthcare workers
New Minibadge expands intuitive mobile solutions for care team collaboration and safety KALAMAZOO, Michigan, USA, March 1, 2022 — Vocera, now part of…
Fiducia’s Inaugural Fund Launched With Initial Size of 3.3 Billion Yen; Fundraising to Continue
TOKYO, Feb. 21, 2022 — Fiducia Inc. announced that it has executed the first closing of its inaugural fund (Fiducia Growth Tech Limited Liability Investment Limited Partnership), with an initial…
The Johns Hopkins University’s Institute for Global Tobacco Control updates its free online course on the dangers of tobacco use and COVID-19
BALTIMORE, Feb. 18, 2022 — With the COVID-19 pandemic still a concern as 2022 begins, arm yourself with knowledge about the connection between tobacco use and COVID-19 with a free online…